Well-known for its use as a steel alloy, vanadium has grown in popularity as a battery metal in recent years. Rechargeable batteries are becoming the fuel for modern life. Electric vehicles, smartphones, tablets, laptops and wind and solar power all depend on these batteries–and we depend on these technologies.
Vanadium
Vanadium (V) is a hard but malleable silver-grey metal. It rarely occurs in nature in its pure metallic form, but is commonly found as a compound in other minerals. Vanadium-bearing minerals, such as vanadinite (Pb5(VO4)3Cl), patronite (VS4) and carnotite (K2(UO2)2(VO4)2·3H2O) have been mined historically. These minerals are rare and do not exist in economic quantities. The majority of vanadium is mined from vanadium-bearing titaniferous magnetites (Fe(Fe,Ti)2O4). Vanadium is extracted from deposits as V2O5 (vanadium pentoxide).

Uses of Vanadium
The majority of produced vanadium–85%–is used as a steel additive. Ferrovanadium (FeV) is an alloy added to steel to strengthen and harden the material. Vanadium is resistant to corrosion and also acts as an anti-corrosive when added to steel. Vanadium-enriched steel is used to make hand tools, pipes, rebar, automotive parts and more. When alloyed with titanium, vanadium provides temperature stability and strength. This material is used in jet engines, aircraft frames and dental implants.
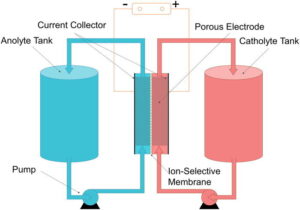
Originally developed by researchers at the University of New South Wales, vanadium redox flow batteries have been used commercially since the mid-2010s. These batteries pump electrolytes dissolved in liquids through electrochemical cells separated by a membrane. Vanadium exists in four oxidation states, so vanadium pentoxide (V2O5) dissolved in sulfuric acid can be used in both the positive and negative electrolyte tanks. Vanadium redox batteries are long-lasting, have a very large storage capacity, a rapid response time, and can be stored unused for long periods of time. They are ideal for large fixed applications, such as grid energy storage for wind and solar power operations. Vanadium is also used in glass coating and ceramics manufacturing, and as a catalyst.
Vanadium Deposit Styles
Vanadium is typically found in three types of deposits: vanadiferous titanomagnetite (VTM) deposits, sandstone-hosted vanadium (SSV) and vanadium-rich black shales.

- VTM deposits: magmatic deposits of titanomagnetite ((Fe(Fe,Ti)2O4)) enriched in vanadium. These deposits often occur in mafic or ultramafic igneous rocks (gabbro or anorthosite) and are commonly found in Large Igneous Provinces. Examples of VTM deposits include the Bushveld Complex in South Africa, the Panzhihua layered intrusion in China, the Kachkanar massif in Russia, the Windimurra igneous complex in Australia, the Maracas Menchen mine in Brazil, and the Matagami deposit and the Lac Dore complex in Canada. VTM deposits are typically associated with iron, chromium, copper, nickel or PGE deposits, so vanadium is usually mined in conjunction with these metals.
- Sandstone-hosted vanadium: vanadium and uranium enriched lenses within sandstone bodies. The most significant SSV deposit is in the Colorado Plateau in the United States, which were originally mined for uranium with vanadium as a byproduct. SSV deposits are also present in Australia, Argentina and Kazakhstan.
- Vanadium-rich black shales: organic-rich marine shales enriched in vanadium. These vanadium deposits are commonly associated with marine oil shales and phosphorite deposits. Vanadium deposits in black shales are known to occur in the United States (Nevada, Idaho, Wyoming, Illinois, Indiana and Montana), Australia, Madagascar, Colombia and Venezuela. None of these deposits are being exploited for vanadium, although projects are underway in Nevada (Gibellini) and Australia (Julia Creek). Vanadium-rich black shale deposits often contain high amounts of other metals, including molybdenum, nickel, PGEs, zinc, uranium and phosphorus.

Less common vanadium sources include:
- Vanadate deposits: deposits of vanadates (minerals consisting of vanadium, oxygen and other metals such as zinc, lead and copper). Vanadates typically form in the oxidized zones of base metal deposits. The copper-lead-zinc vanadate deposit in northern Namibia used to be considered the world’s largest vanadium deposit. However, the mine closed in 1978. Vanadate deposits exist in Angola, South Africa, Zambia, Zimbabwe, Argentina, Mexico and the United States, but are not economically significant.
- Byproduct of petroleum, coal and tar sands: petroleum extracted from vanadium-rich organic shales can contain up to 1400 ppm of vanadium. Vanadium is separated from the crude oil during the refining process. Vanadium-rich oil fields, tar sand deposits and coal deposits are found in Venezuela, the United States, Canada, Iran and Japan.
- Cobalt-rich manganese crust: deposits of metallic minerals (cobalt, vanadium, molybdenum, platinum, and tellurium) in the ocean. Cobalt-rich manganese crust forms deep in the oceans, on the flanks of seamounts. The crust is made of minerals precipitated from the cold ocean water. The Clarion-Clipperton zone in the Pacific Ocean is currently being explored as a potential project for deep-sea vanadium mining.
Global Supply and Future Outlook
The bulk of the world’s vanadium–55%–comes from China. Significant production also comes from Russia, Brazil and South Africa. Demand is largely for construction materials (steel) and is concentrated in China. However, vanadium demand may increase if vanadium redox flow batteries grow in popularity. Governments are increasingly looking towards clean, renewable sources of energy like wind and solar. Vanadium redox flow batteries could be the key to integrating renewable energy sources with existing power grids.
Sources
Notable Vanadium Projects
Subscribe for Email Updates