Laterite is formed when heavy tropical rainfall results in intense weathering of rock and soil. After many millions of years what is left is called a “laterite”, which can be either iron-rich, or in extreme cases, aluminium-rich
Most of the world’s aluminium is mined from lateritic bauxite deposits. The largest producer is Australia’s Weipa Mine in northern Queensland, while other countries with large reserves include Guinea, Vietnam, and Jamaica.
Formed in a similar manner to iron laterites, bauxite is created by weathering of suitable rocks in a tropical climate. Typically deposits are large and at the surface. Mining usually involves simple strip-mining, without the need for open pit development.
Lateritic bauxite formation
Laterite is formed when heavy tropical rainfall results in intense weathering of rock and soil. The chemistry of weathering silica-rich rocks sees the relatively mobile elements such as calcium, sodium, potassium and magnesium being washed away, while the immobile metals including aluminium, iron, titanium, and zirconium remain.
After many millions of years what is left is called a “laterite”, which can be either iron-rich, or in extreme cases, aluminium-rich (i.e. bauxite).
Most lateritic bauxites were formed in a period from the mid-Cretaceous to the late-Tertiary, that is, 100 million to 2 million years ago. During this time, laterite formation was not continuous. Instead, they formed during relatively short periods of intense weathering separated by long periods of less intense weathering.
Many lateritic bauxites are pisolitic (Figure 1). The word pisolitic comes from the Greek meaning pea-sized rock particles. When cut or broken open their inside surfaces reveal concentric bands of different mineral compositions.

Mineralogy of lateritic bauxite
The basic mineralogy of lateritic bauxites is similar throughout the world. The most sought after bauxite mineral is the aluminium hydroxide mineral gibbsite, Al(OH)3. Although it has a lower aluminium content than some other minerals, gibbsite-rich bauxite is cheaper to mine and process. Other aluminium minerals which may be present in bauxite include diaspore, boehmite (AlO(OH)) and various types of alumina (or Al2O3).
Accessory minerals associated with lateritic bauxite include; kaolinite, hematite, goethite, anatase, zircon and unweathered quartz.
The lateritic bauxite profile
A “typical” profile is really a misnomer as there are so many variations. The example shown in Figure 2 is a lateritic bauxite profile exposed in the western coastal cliffs of Cape York Peninsula, in northern Australia. Elements of this “typical” profile are seen in most lateritic bauxite profiles in other parts of the world.
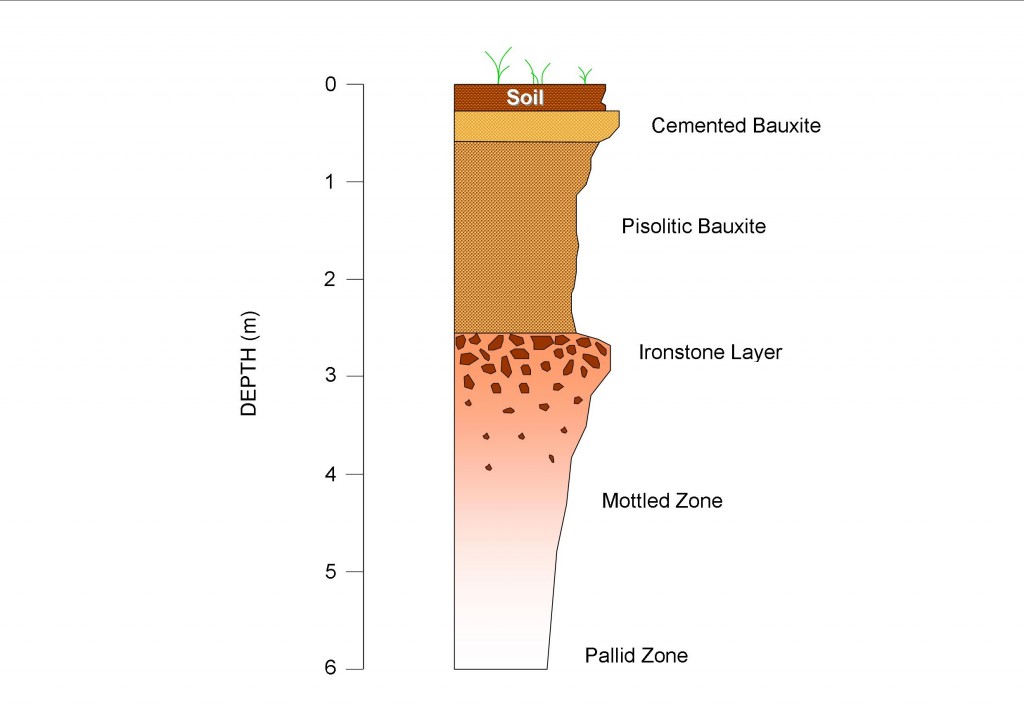
The profile consists of a relatively thin soil layer overlying a horizon consisting of cemented pisolitic bauxite. The main mineral within this cemented layer is boehmite or “monohydrate” as the miners call it. Although one of the most aluminium-rich minerals in lateritic bauxite, the layer containing this mineral is normally stripped off and discarded during mining. This is because the layer is typically cemented and would therefore require crushing. In addition, the caustic soda solution that is used to extract aluminium from bauxite needs to be much hotter for boehmite than for gibbsite, so processing costs would be correspondingly higher.
Underlying the cemented bauxite is a layer of loose pisolitic bauxite 1-2m thick. The main mineral in the pisolitic bauxite is the aluminium-rich mineral gibbsite or “trihydrate” as it is commonly referred to. Pisolitic bauxite ore is an ideal ore for various reasons. It is located close to the surface so very little cover is needed to be removed to expose it. The bauxite is loose and easily mined with front-end loaders. Beneficiation of the ore before it is shipped out of port involves simple screening and washing. Selective mining ensures that only the highest grades are mined.
Below the pisolitic bauxite horizon is a nodular ironstone layer. At this level, the bauxite becomes increasingly iron and kaolin-rich. Kaolinite, which contains a great deal of silica, is also much lower in aluminium content than bauxite. Kaolinite tends to dissolve relatively easily in the caustic soda solution during aluminium extraction so uses up this valuable chemical. For this reason, mining has to be done very carefully as the ironstone layer is approached because kaolinite in the ore significantly reduces its value.
Beneath the ironstone layer the profile becomes increasingly less nodular and more mottled in appearance. The mottled zone has reddish coloured patches containing hematite and goethite, within a pale coloured kaolin-rich mass. At greater depths below this zone the white mineral kaolinite dominates, hence the name pallid zone given to the lowest part of the weathering profile.