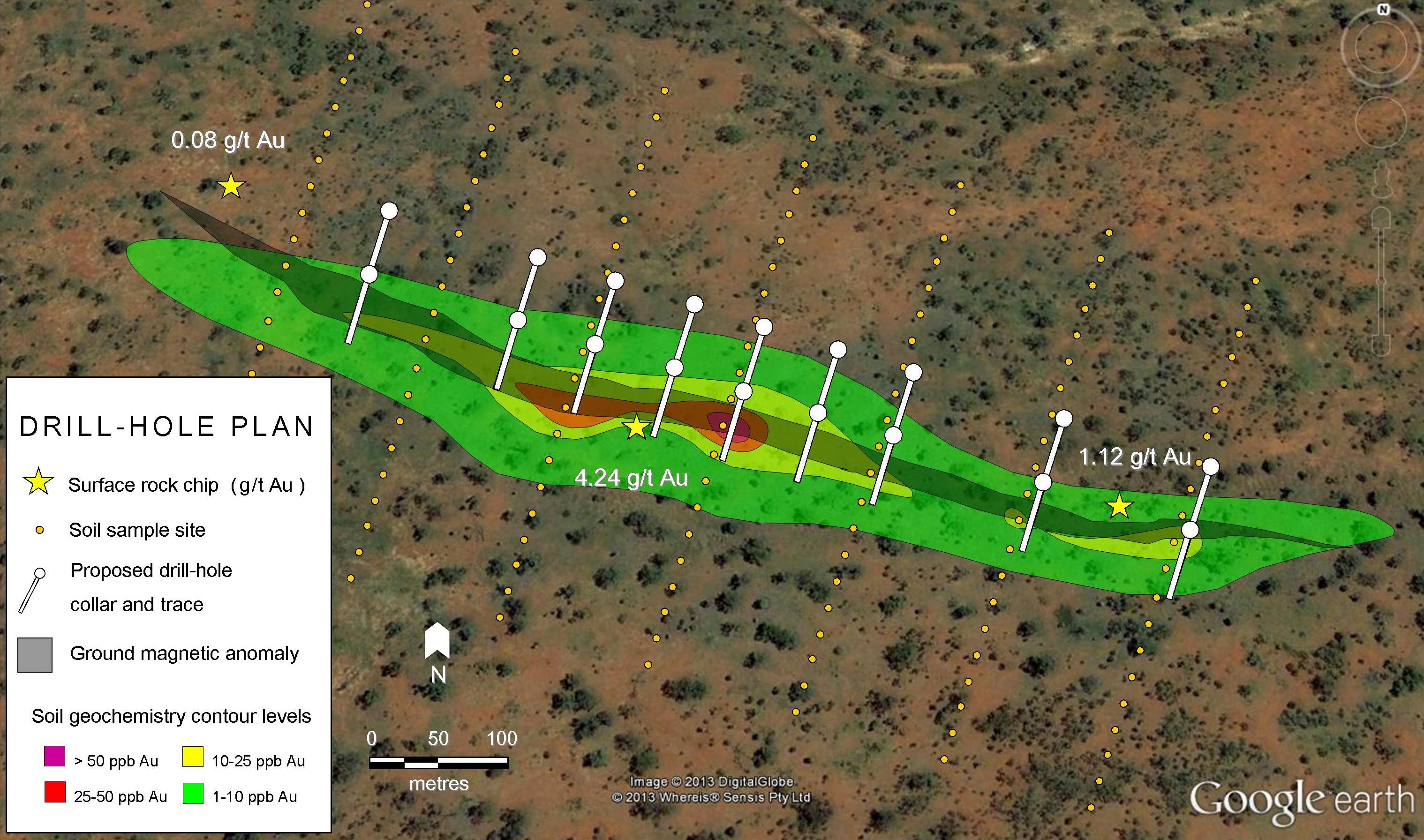
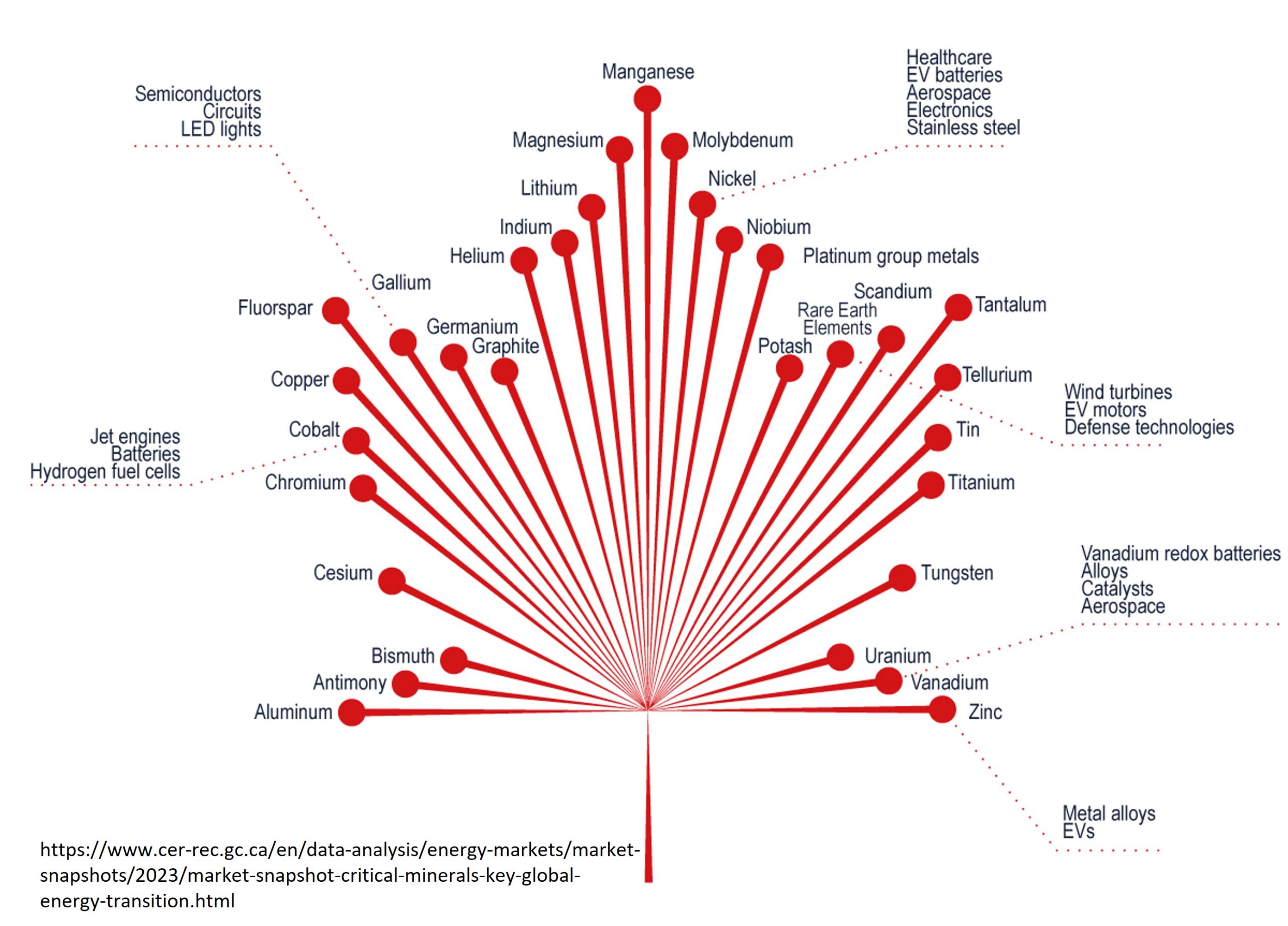
Zinc is the 30th element on the periodic table. It has the symbol Zn, which is an abbreviation of zinke; the German word for “tooth-like, pointed or jagged”, which probably refers to the needle-like crystals when molten zinc solidifies and crystallises.
Zinc is hard and brittle at most temperatures and has a relatively low melting point of 420°C. Zinc is just over seven times heavier than the same volume of water.
Zinc is commonly found in association with copper and lead in various ores. The main zinc ore mineral is called sphalerite, which is a zinc sulphide
What Are The Main Uses For Zinc?
Zinc is the fourth most common metal in use after iron, aluminium and copper.
The main use for zinc is in galvanising iron or steel, which is a form of rust proofing. This works because zinc is more chemically reactive than iron or steel. Galvanising involves dipping sheets of iron or steel into molten zinc or by a process of electroplating. When removed from the zinc bath, the zinc solidifies and crystallises producing a typical feathery pattern. The zinc coating protects the underlying metal from corrosion because it attracts corrosion instead of the underlying metal. The term sacrificial metal is used for any metal which has this ability. Protection is maintained even if the coating is scratched or abraded, unlike coatings of paint and plastic. The eventual formation of a zinc carbonate layer from the corrosion of the zinc coating has a protective effect as well.

The use of zinc as a sacrificial metal can be applied to protecting submerged and buried iron and steel structures in another way. Instead of coating the iron or steel, pieces of zinc are bolted to the structures or connected via a wire. As the zinc corrodes, the iron or steel structure is protected from rusting due to the electrical circuit that is set up between the two metals.
Another important use is in the production of various kinds of alloys for use in die-casting. The most well known alloy is brass, which has been produced since the 10th century BC. Normal brass contains from 30 to 40 percent zinc, however depending on the hardness and other properties required proportions outside this range are also used. Like bronze, brass is harder and tougher than pure copper.
Zinc metal is used in electric batteries as the anode. In traditional dry cells the container is made of zinc.
Zinc is in also used in the extraction of gold. Zinc shavings and powder precipitate gold from a cyanide solution, commonly used to leach gold from ore.
A variety of chemical compounds with numerous uses are produced from zinc. These range from industrial compounds to health care products. For example, zinc oxide and zinc sulphide are used as pigments in paint and zinc oxide is used in some sunscreens. Dietary mineral supplements are another important use for zinc.

How Are Zinc Ore Deposits Formed
Zinc is the 24th most abundant element in the Earth’s crust. It is commonly found in association with copper and lead in various ores. The main zinc ore mineral is called sphalerite, which is a zinc sulphide. Sphalerite is also known as zinc blende and contains 67% zinc when pure. It is commonly associated with galena, a lead sulphide mineral. The main deposit type that is mined for zinc is silver-lead-zinc strataform and strata-bound deposits.
The three top producing countries of zinc in the world are China, Peru and Australia. These are followed by India, USA and Canada.