Many civilisations have had an eternal love affair with gold. As a consequence, wars have been fought over it and many lives have been lost. It has become a symbol of wealth and power in many countries of the world. It is traded as a currency and is viewed as a safe haven in times of economic uncertainty. Gold is number 79 on the periodic table of the elements. It has the symbol Au, which is an abbreviation of aurum, the Latin word for gold. It is in the same chemical group as silver and copper, and therefore shares some similar properties, such as a high electrical conductivity, malleability, and resistance to oxidation.
What are its uses?
Gold is prized for its bright yellow colour and metallic lustre; its resistance to tarnishing in water and air; its softness and ease of working into intricate designs; and its scarcity. All of which has made gold an ideal metal for jewellery making and ceremonial items. In fact, gold is too soft in many cases and must be alloyed with copper and silver to increase its toughness and resistance to wear. Nine carat gold (9ct) contains only 37.5% gold whilst 18 carat gold (18ct) consists of 75% gold. In many parts of Asia, 24 carat or pure gold is the only type of gold jewellery worn despite its softness.
Gold is heavy, with a specific gravity of 19.3 times that of water. The old timers made use of this property when separating gold particles from sand and gravel in placer deposits using pans and sluices.
In the past, it was common to have high denomination coins minted out of gold. The milled edge around coins is there for a reason. It was to help stop the illegal practice of shaving the edges of coins to obtain “free” gold. The milled edge is still used on coins today even when made from non-precious metals. This gives them the appearance of value. As time passed the value of gold contained within the coin eventually surpassed its face value by hundreds of times. This resulted in gold coins being sold as bullion or melted down into ingots.
With the advent of electronics last century the use of gold in electrical circuitry and wiring has becoming increasingly important, particularly as integrated circuits are miniaturised to fit into smaller and smaller devices. The ability to draw gold out into extremely thin wire and its resistance to oxidation makes it ideal for this purpose.
One other use for gold is in dentistry where it is used for fillings.
Where is it found?
Gold is found in many different mineral deposit types and settings. These include epithermal, mesothermal, porphyry, volcanogenic massive sulphide (VMS), iron oxide copper gold (IOCG) and placer deposits, just to name a few.
Coarse-grained gold is commonly found in association with quartz (Figure 1). Quartz veins containing gold were targeted by old time prospectors as they were relatively easy to spot while walking across the land.
Gold can combine with sulphides, such as pyrite and arsenopyrite, in the form of extremely fine micron-sized grains. When in this form, metallurgists often have difficulty in recovering all the gold from the ore.
In the Victorian goldfields of Australia, extremely large nuggets are found in near surface deposits. These can be several kilograms in weight and are thought to have formed in situ by the transport and deposition of gold in suspension.
“Gold is where you find it!”
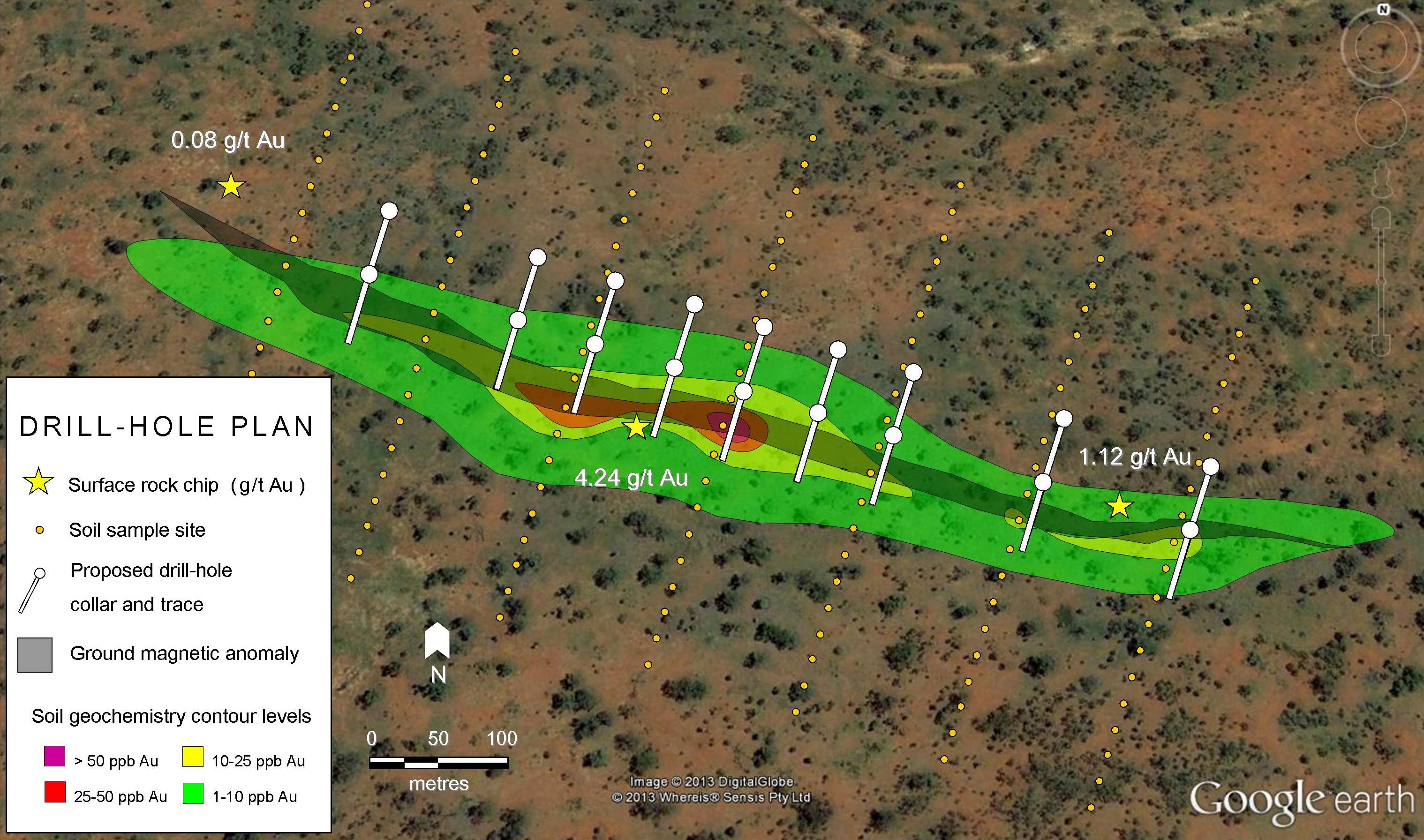
There is phrase often used in gold exploration; “Gold is where you find it!” Its location within deposits can be quite elusive at times. Campaign drilling of a mineralised shear or vein can often miss high grade shoots due to their narrowness, much to the disillusionment of the geologist in charge.

Figure 1: Gold within quartz.