Tin is number 50 on the periodic table of the elements. It has the symbol Sn, which is an abbreviation of stannum; the Latin word for tin. With a concentration within the earth’s crust of just 2 parts in every million, or 2 ppm, it is not a common element.
Tin shares similar properties to germanium and lead, which are located nearby in the periodic table. Tin has a low toxicity, which is a big advantage over lead. The metallic form of tin, is known as white tin or beta-tin, and has a relatively low melting point of around 232°C.
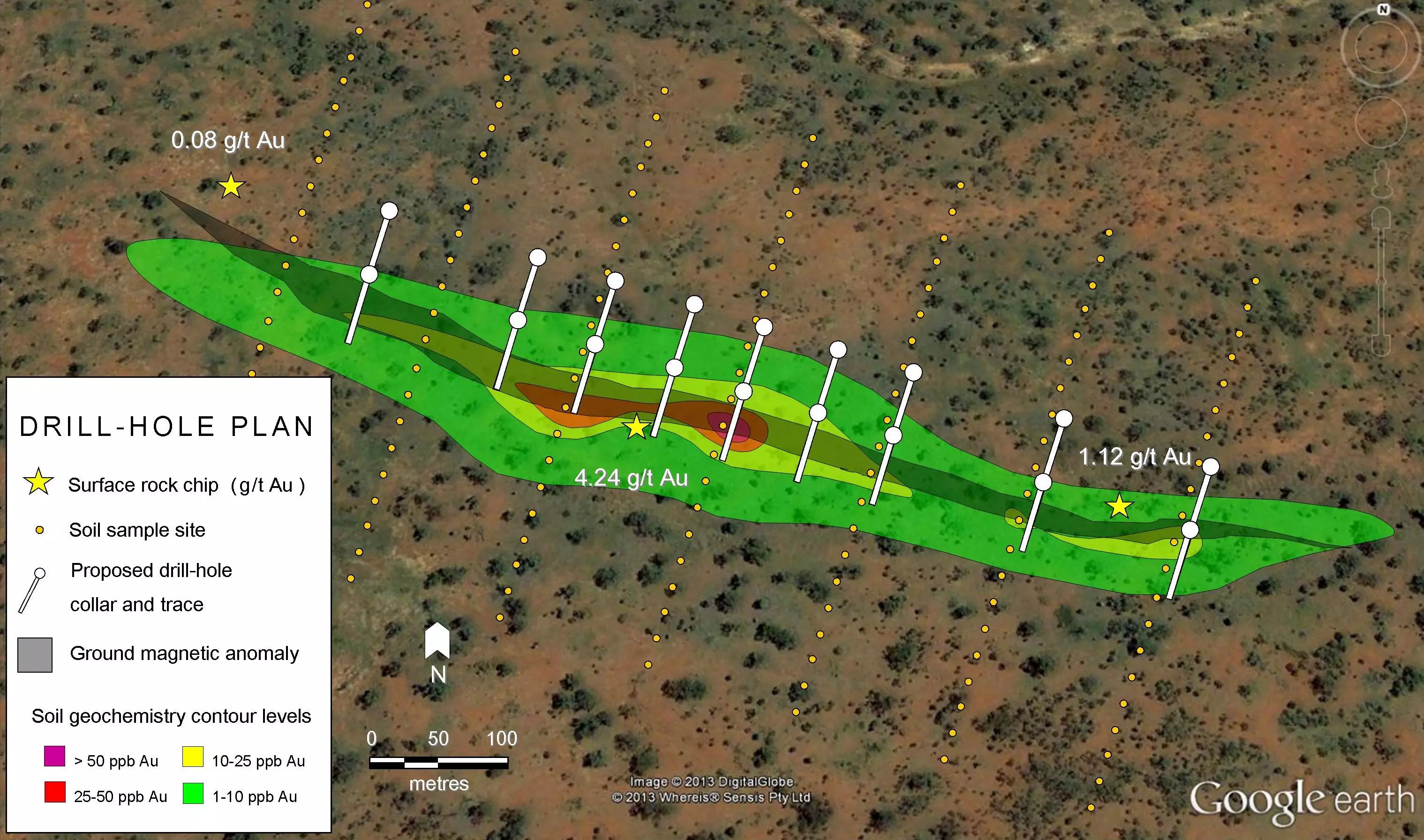
The majority of the world’s production of tin, around 80%, is produced from placer, particularly alluvial, deposits.

White tin is a silvery-white, ductile and malleable metal. It is not easily oxidised in air so is used to prevent corrosion on other metals such as steel.
Tin is found in another form, called grey tin or alpha-tin. Grey tin has a similar crystalline structure to diamond but is not as strong and is quite brittle. The two types of tin are known as allotropes, that is, they are the same element except with different crystal structures.
Pure tin has an undesirable property known as tin pest, where the metallic form transforms into the non-metallic form, at temperatures below 13°C. The addition of impurities in the tin, such as bismuth, antimony, lead and silver can suppress this transformation to a lower temperature or may even prevent it from happening.
What Are The Main Uses For Tin?
The non-metallic grey tin has no common uses apart from some specialised applications in semi-conductors. The metallic white tin on the other hand has a variety of uses.
Since 3000 BC, tin has been used in bronze where it is alloyed with copper. It wasn’t until after 600 BC that the pure metallic-form of tin was produced.
Since the Bronze Age, tin has been used in pewter for dinnerware. Pewter is an alloy composed of 85-90 % tin with some copper, antimony and/or lead.
Solder is used for joining metal parts together, such as on electronic circuit boards. Soft solders were traditionally composed of tin and lead. Due to health concerns the lead component of solders is now being removed and replaced by other metals.
The main modern day use of tin is for plating or coating steel to prevent corrosion. Its low toxicity makes tin-plated steel useful in food packaging, such as the common tin can.

An indirect, but very important, use for tin is in the manufacture of glass sheets used in windows. In the Pilkington process, molten glass is floated onto a bath of molten tin. The glass flows over the tin producing a perfectly flat surface. Tin is used because of its relatively low melting point, low reactivity with the glass and high specific gravity compared to the glass, which ensures that the glass floats on top of the molten tin..
Where is Tin Found?

The most economically important tin mineral is cassiterite, a mineral composed of tin and oxygen. Cassiterite can be found in primary ore deposits within veins and quartz lodes with granite or at the margins of granite bodies where they are in contact with sedimentary rocks. Cassiterite is extremely resistant to both chemical and physical weathering, so tends to form placer deposits as it’s concentrated by the action of water and other agents. The majority of the world’s production of tin, around 80%, is produced from placer, particularly alluvial, deposits.
Tin is also found in the mineral stannite, a tin-copper-iron sulphide. However, stannite is not normally sought after as an ore mineral due to extract difficulties.
China, Indonesia and Peru are currently the world’s largest producers of tin. It is expected that prices will continue to rise due to increasing demand and reducing known reserves of this metal. Throughout the world, more and more tin is being recycled to meet some of this growing demand.