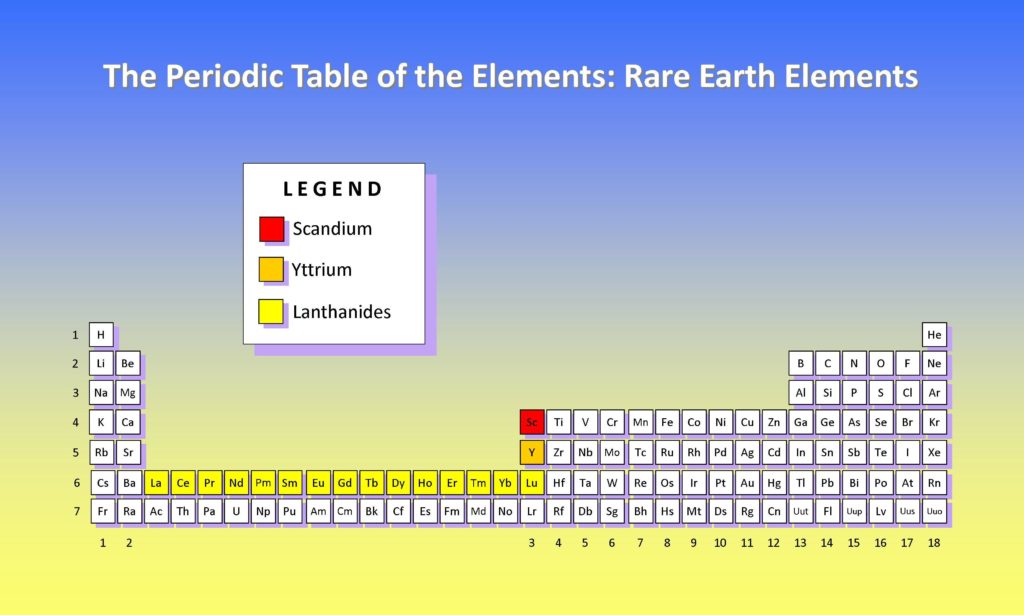
There are 17 rare earth elements or REE for short. They include the 15 lanthanides, plus scandium (Sc) and yttrium (Y). The lanthanides are range from the lightest lanthanum (La), cerium (Ce), praseodymium (Pr), through to the heavier REE’s including ytterbium (Yb) and lutetium (Lu).
The term “rare earth element” is really a misnomer. Apart from the exceedingly scarce and radioactive promethium, most of the rare earth elements are relatively abundant in the earth’s crust.
The term “rare earth element” is really a misnomer. Apart from the exceedingly scarce and radioactive promethium, most of the rare earth elements are relatively abundant in the earth’s crust. The reason for the name is that they are highly reactive and only rarely found concentrated into mineral deposits. Rare earth mineral deposits normally contain a mixture of the element’s , because of their generally similar chemical properties. In addition, lighter rare earth’s, e.g. cerium, tend to coexist separately from the heavier elements, e.g. yttrium.
What are their uses?
Although rare earth elements have similar chemical properties, each rare earth element has enough unique characteristics, for their use in a wide variety of different applications.
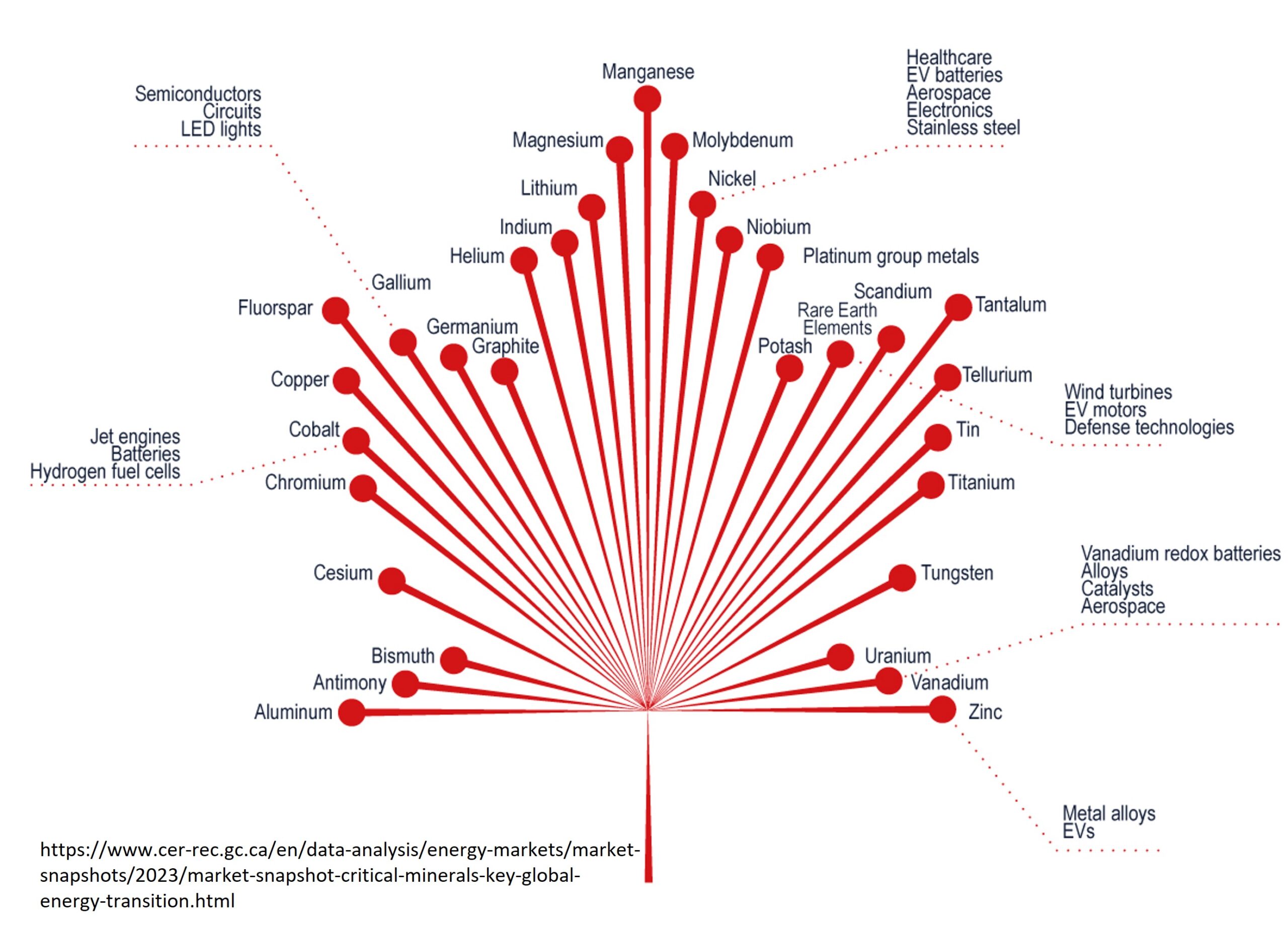
Rare earth elements are critical in the manufacture of a number of high tech products. For example, they are used in magnets, catalysts, phosphors, glass, polishing compounds, metal alloys and ceramics, just to name a few.
Neodymium is alloyed with iron and boron to produce powerful permanent magnets. These are used increasingly in portable electronic equipment, particularly in loudspeakers and computer hard drives. Their high efficiency makes them useful in hybrid cars and wind turbines.
Lanthanum is an additive in the manufacture of high refractive index glass, used in telescope and camera lenses. Lanthanum also has a role in crude oil refining as a petroleum cracking catalyst.
Cerium too is used in oil refining and also in vehicle catalytic converters to reduce harmful emissions. Cerium oxide is a polishing compound for glass.
Praseodymium is a constituent in special glass for making visors for protecting welders and glass makers.
Yttrium, terbium and europium are increasingly used in television and computer screens and other visual display devices. They are known as phosphors which emit light of certain colours when irradiated with particles of electromagnetic radiation.

Where are they found?
Most of the world’s supply of lanthanides are derived from the mineral bastnäsite (or bastnaesite) which is a lanthanide fluoro-carbonate mineral found in certain types of granite and associated pegmatite veins, as well as in carbonatite, an igneous rock containing greater than 50% carbonate minerals.
There are three types of bastnäsite; bastnäsite-Ce, bastnäsite-La and bastnäsite-Y. The different suffixes indicate the predominant rare earth element present within each type.
Monazite, a rare earth phosphate mineral is another source of rare earth elements. There are four main varieties of monazite: monazite-Ce, monazite-La, monazite-Nd and monazite-Sm. Monazite may also contain thorium making it radioactive. Monazite occurs as a rather rare accessory mineral in certain granitic rocks and pegmatite veins. More commonly, it is concentrated in stream and beach sands. Unlike bastnäsite, monazite tends to have a full range of rare earth elements, from the lighter ones to the heavier ones. Bastnäsite, on the other hand, contains mostly lighter rare earth elements. Despite this, the radioactive nature of monazite has made bastnäsite the preferred source of rare earth elements. The disposal of radioactive material, a by-product from mineral processing, is an issue that needs to be resolved whenever monazite is used.
China currently has a virtual monopoly on the production of rare earth elements, accounting for nearly all of the world’s supply of this important commodity. In China, rare earths are found in relatively uncommon “ion absorption clay” ore deposits. In comparison, the next biggest producers, USA and India, produce only a few percent of the total world’s production between them. With demand escalating, due to increasing use in high tech industries, prices for the rare earths should continue to rise.